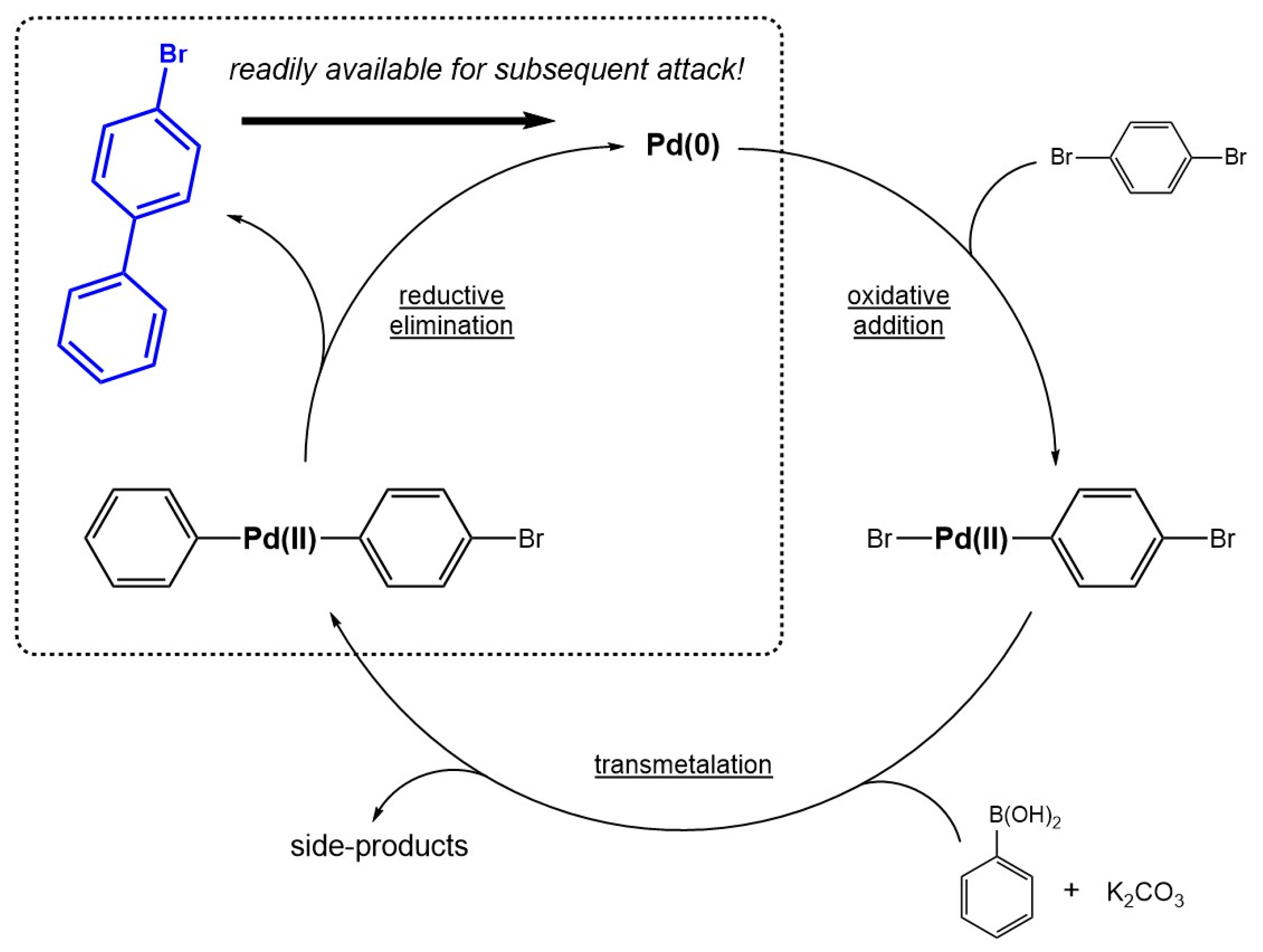
Catalysts | Free Full-Text | Highlights on the General Preference for Multi-Over Mono-Coupling in the Suzuki–Miyaura Reaction

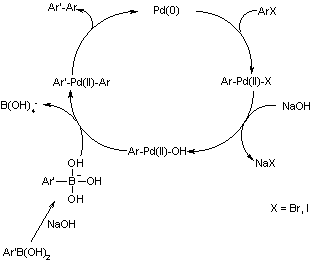
Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library

Suzuki-Miyaura Cross-Coupling Reaction and Potential Applications: Kostas, Ioannis D: 9783038425564: Amazon.com: Books

Palladium-Catalyzed Decarbonylative Suzuki–Miyaura Coupling of Amides To Achieve Biaryls via C–N Bond Cleavage | The Journal of Organic Chemistry

Suzuki−Miyaura coupling and O−arylation reactions catalysed by palladium(II) complexes of bulky ligands bearing naphthalene core, Schiff base functionality and biarylphosphine moiety - ScienceDirect

Silk−Fibroin‐Supported Palladium Catalyst for Suzuki‐Miyaura and Ullmann Coupling Reactions of Aryl Chlorides - Rizzo - 2022 - European Journal of Organic Chemistry - Wiley Online Library

Palladium-Catalyzed Suzuki−Miyaura Cross-Coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Accounts of Chemical Research

A Bulky Chiral N-Heterocyclic Carbene Palladium Catalyst Enables Highly Enantioselective Suzuki–Miyaura Cross-Coupling Reactions for the Synthesis of Biaryl Atropisomers | Journal of the American Chemical Society

A Bulky Chiral N-Heterocyclic Carbene Palladium Catalyst Enables Highly Enantioselective Suzuki–Miyaura Cross-Coupling Reactions for the Synthesis of Biaryl Atropisomers | Journal of the American Chemical Society

Suzuki-Miyaura Cross-Coupling Reaction and Potential Applications: Kostas, Ioannis D: 9783038425564: Amazon.com: Books

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society

Palladium catalyzed asymmetric Suzuki–Miyaura coupling reactions to axially chiral biaryl compounds: Chiral ligands and recent advances - ScienceDirect

Palladium-Catalyzed Suzuki–Miyaura Coupling of Aryl Esters | Journal of the American Chemical Society

A Convoluted Polyvinylpyridine‐Palladium Catalyst for Suzuki‐Miyaura Coupling and C−H Arylation - Ohno - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library

Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library

Mechanistic Aspects of the Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling Reaction - D'Alterio - 2021 - Chemistry – A European Journal - Wiley Online Library
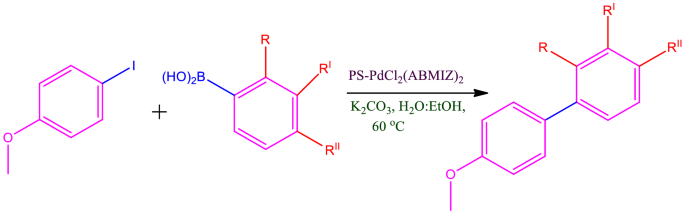
Suzuki–Miyaura Cross Coupling Reaction Using Reusable Polymer Anchored Palladium Catalyst | Catalysis Letters

Homogeneous and Recyclable Palladium Catalysts: Application in Suzuki– Miyaura Cross-Coupling Reactions | Organometallics
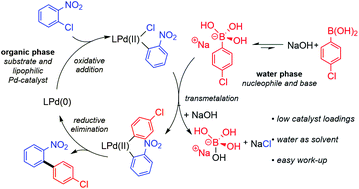
Pd-Catalysed Suzuki–Miyaura cross-coupling of aryl chlorides at low catalyst loadings in water for the synthesis of industrially important fungicides - Green Chemistry (RSC Publishing)